SIGNAL-PROCESSING IN THE BRAIN
"Method to Generate Self-Organizing Processes in
US
US 6172941 (filing date Dec. 16, 1999) Author:
Erich
Bieramperl, 4040 Linz, Austria
(b) If the threshold P1 is exceeded, then an "action potential" AP (as in Fig. 4a) is produced
whichpropagates actively along the cell membrane (or axon fiber) with a constant amplitude in a self-
regenerating manner. The production of AP is akin to a spark incident at a blasting fuse: the fiercely
burning powder heats neighboring parts of the fuse, causing the powder there to burn, and so on, thus
propagating the flame along the fuse.
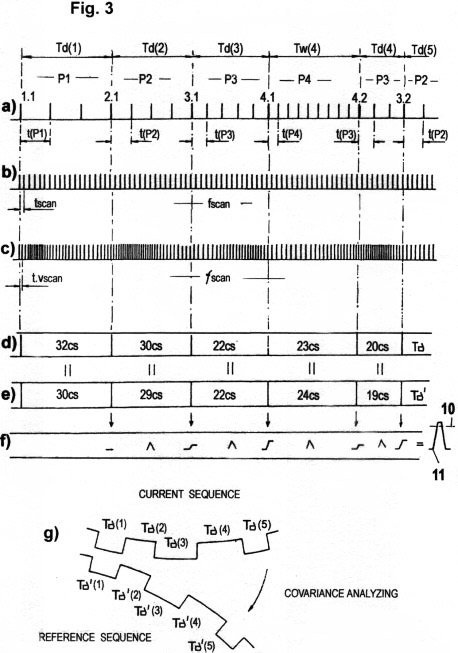
AP's are used in the quantization of STQ(d) and STQ(v) elapse times. They are
practically equivalent to identification pulses IP with periods t(P1),
t(P2), t(Pn)..., which are shown in Fig. 3a. AP's signal the occurrence
of the phase transitions from which STQ(d) and STQ(v) elapse times
derive. In addition,the AP' indirectly activate the molecular/biological "timers" that are used for
recording these elapse times. But AP's do not represent deterministic
sampling rates for amplitude scanning; and they do not correspond to
electronic voltage/frequency converters. Moreover, their amplitude is
independent of the stimulation intensity at the receptor, and they do
not represent the time counting pulses used in the measurement of elapse
times. Rather, the recording of STQ elapse times is effected and
modulated by the velocity with which the action potentials propagate
along the nerve fibers (axons) and membrane regions.
see:
Excerpt of Patent
Description:
Autonomous Mechanisms and Organisms"
(pages 9-17)
EP EP 01145406 A1 (filing
date Dec. 03,1999)
Figs. 4a - d illustrate a model for the acquisition and processing of STQ(d) and STQ(v) elapse
times (see also Figs. 3a-g) and for temporal and motoric auto-adaptation in a molecular/biological context.
The basic elements of the model have already been described in the neurophysiology literature by
KATZ, GRAY, KELLY, REDMAN, J. ECCLES and others. The present invention is of special
originality because temporal and motoric auto-adaptation is effected here by means of STQ quanta,
which are described for the first time here. Such systems consist mainly of numerous neurons (nerve
cells). The neurons are interconnected with receptors (sensory neurons), which enables the recording
and recognition of the neurons' physical surroundings. In addition, the neurons cooperate with effectors
(e.g.muscles) which serve as command executors for the motoric activity. The expression "receptor"
or "sensory neuron" corresponds to the mechanistic term "sensor". An "effector" is the same as an
"actuator", which is a known term in the cybernetics literature. Each neuron consists of a cell membrane
that encloses the cell contents and the cell nucleus. Varying numbers of branches from the neurons
(axons, dendrites etc.) process information off to effectors or other neurons. The junction of a dendritic
or axional ending with another cell is called a synapse. The neurons themselves can be understood as
complex biomolecular sensors and time pulse generators; the synapses are time data analyzers which
continually compare the currently recorded elapse time sequences with prior recorded elapse time
patterns that were produced by the sensory neurons and were propagated along nerve fibers towards the
synapses. In turn, a type of "covariance analysis" is carried out there, and adequate probability density
signals are generated that propagate to other neighboring neural systems or to effectors.
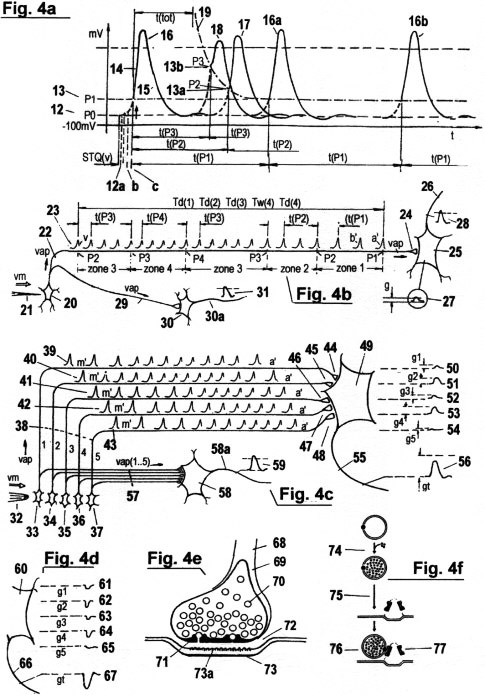
Fig. 4a shows a so-called "action potential" AP that is produced at the cell membrane by an
abrupt alteration of the distribution of sodium and potassium ions in the intra and extra-cellular
solution, which works like a capacitor. These ionic concentrations keep a certain balance as long
as no stimulus is produced by the receptor cell. In this equilibrium state, a constant negative
potential 12, termed the "rest potential", exists at the cell membrane. As soon as a receptor
perceives a stimulus from an externalsignal source, Na+ ions flow into the neutral cell, which
causes the distribution of positive and negative ions to be suddenly inverted, and the cell
membrane " depolarizes". Depending on the intensity of the receptor stimulus, several effects are
produced:
a) If the threshold P1 is not exceeded, then a so-called "electrotonic potential" EP is produced
which propagates passively along the cell membrane (or axon fiber), and which decreases exponentially
with respect to time and distance traveled. The production of EP is akin to igniting an empty fuse cord.
The flame will stretch itself along the fuse, becoming weaker as it goes along, before finally going out.
EP's originate with each stimulation of a neuron.
The time measuring properties of AP's are described in detail in the following section:
If an EP, in answer to a receptor stimulus, exceeds a certain threshold value (P1) 13,
then an AP is triggered. The amplitude trace of an AP begins with the upstroke 14 and ends
with the repolarisation 15, or with the so-called "refractory period", respectively. At the
end of this process, the membrane potential decreases again to the resting potential P0, and
the ionic distribution returns to equilibrium. Not each receptor stimulus generates sufficient
electric conductivity to produce an AP. As long as it remains under a minimal threshold
value P1, it generates only the electrotonic potential EP (introduced above). (For a better
understanding of elapse time measurements in biological/chemical structures, see Fig. 2c and
Fig. 3a). The first AP, which is triggered after a receptor is stimulated, generates initially
(indirectly) the impulse that activates the first timer that records the first STQ(d) elapse
time, when the signal amplitude W passes through the threshold value of the potential P1 at
phase transition iTw(1.1). This signal represents simultaneously an identification pulse IP.
The first AP corresponds to the first IP in a sequence of IP's that represents the respective
threshold value status or perception zone in which the stimulation amplitudes were just found.
As long as the stimulus at the receptor persists, an AP 16a, 16b... is triggered in temporal
intervals whose duration depends on the respective thresholds in which the stimulus intensities
have just been found.
These temporal intervals correspond to those IP periods t(P1), t(P2),... that
are required for serial
allocation and processing of STQ elapse times (see Fig. 3a). The AP
frequency is stabilised through the
so-called "relative refractory period" (i.e. downtime) after each AP,
during which no new depolarisation is possible. Because the relative
refractory period shortens itself adaptively in proportion to the
increase in stimulation intensity at the receptor (e.g. if the EP
reaches a higher threshold value P2 (or perception zone) 13a),
there is a similarity here with "programmable bi-stable multivibrators"
found in the usual mechanistic electronics. The downtime (refractory
period) after an AP is shown as the divided line 19.
Fig. 4a illustrates an "absolute refractory period" t(tot) following a
repolarisation. No new AP can be created during this time, irrespective of the
stimulation intensity at the receptor rises. The maximum magnitude of a
recognizable receptor stimulus is programmed in this way. Of importance is the
fact that both the duration of the relative refractory period as well as character of
the absolute refractory period are subordinate to auto-adaptive
regularities, and are therefore continually adapting to newly appearing
conditions in the organism. Consequently, the threshold values P0, P1,
P2.... from which STQ quanta are derived are themselves not absolute
values, but are subject to adaptive alteration like all other
parameters; including, in particular, the physical "time".
We shall now elaborate upon what happens after the first STQ(d) elapse time at P1 is recorded
via the first AP: If the stimulation intensity (with a theoretical amplitude W) increases from
the lower threshold P1 to the next higher threshold P2, then the following AP triggers
indirectly the recording of the second STQ(d) elapse time as soon as a phase transition
occurs through the next higher threshold P2. The same process is repeated in turn for the
threshold values P3, P4, ... and so on. In each case, the AP functions simultaneously as an
identification pulse IP, as described in Fig. 3a. It therefore recurs in threshold-dependent
periods as long as a perception acts upon the receptor (i.e. for as long as the receptor is
perceiving something).
As an
example, consider also Fig. 3a: As long as the stimulation intensity
remains in the zone P2, the AP 17, 17a, 17b.... recurs in short
temporal periods. These periods (or intervals) are similar to those
periods
of IP identification pulses (with period t(P2)) that are required for
serial recording of the STQ
elapse
times Td(2) and Tw(2). When the increasing stimulation intensity reaches
the threshold value P3
(or perception zone 3) 13b, the AP's recur in even shorter time
periods 18a, 18b, 18c... This
corresponds to the IP identification pulses with the period t(P3), shown
in the figure, which are indirectly required for serial timing of the
STQ elapse times Td(3) and Tw(3). An even larger stimulation intensity,
for
example in P4 (perception zone 4), would generate an even shorter period
for the AP's. This would
correspond approximately to t(P4) in Fig. 3a. The maximum possible AP
pulse frequency is determined by t(tot). Shorter refractory periods,
after the depolarization of APs, also produce smaller AP-amplitudes.
This property simplifies the allocation of AP's in addition. In the
following, the generation of the actual time counting pulses for STQ
quantization is detailed. These pulses are either invariable ITCP or
vm-proportional VTCP, as illustrated in Fig. 3a. The time counting
pulses for the quantization of elapse times are dependent on the
velocity with which the AP propagate along an axon. This velocity is in
turn dependent on the "rest potential" and on the concentration of Na+
flowing into the intracellular space at the start of the depolarization
process, as soon as perception at the receptor cell causes an electric
current to influence the extra/intra-cellular ionic equilibrium.
With the commencement of stimulation of a receptor (at the outset of a
perception), only capacitive
current
flows from the extra-cellular space into the intracellular fluid. This
generates an "electrotonic
potential" EP, which propagates passively. If this EP exceeds the
threshold P1, then an AP, which
propagates in a self-regenerating manner along the membrane districts,
is produced. The greater the capacitive current still available after
depolarisation (or "charge reversal") of the membrane capacitor, the
greater the Na+ ion flow into the intracellular space, and the greater
the available EP current that can
flow
into still undepolarized areas. The rate of further depolarization
processes in the neuronal fibres,
and
consequently the propagation speeds of further AP's, are thus increased
proportionally. The charge reversal time of the membrane capacitor is
therefore the parameter that determines the value 12 of the
resting potential P0. When a stimulus ("excitation") starts from the
lowest resting potential 12,
then
the Na+ influx is the largest, the EP-rise is steepest and the
electrotonic flux is maximum. If an AP
is
triggered, then its propagation speed is in this case also maximum. But
when a receptor stimulus starts
from a
higher potential 12a, 12b, 12c...., then the Na+ influx is
partially inactivated, and the steepness
of the
EP-rise as well as its electrotonic flux velocity is decreased.
Therefore, the propagation speed of an AP decreases too.These specific
properties are used in molecular/biologic organisms to produce
either
invariant time counting impulses ITCP, with periods tscan, or variable
time counting impulses VTCP
with
periods t.vscan. In the latter case, the VTCP's are modulated in
accordance with the relative speeds
vm (via
the STQ(v) parameters), and therefore have shorter intervals (see Figs.
3b, 3c). The STQ(v)-quantum is determined by the deviation of the
respective starting-potential from the lowest resting-potential P0, which serves as a reference value, and is measured by the
duration of the capacitive
charging of a cell membrane when a stimulus occurs at the receptor.
The
duration of the charging is inversely proportional to the velocity of
the Na+ influx through the
membrane channels into the intracellular space. A cell membrane can be
understood as an electric
capacitor, in which two conducting media, the intracellular and the
extracellular solution, are separated from one another by the
non-conducting layer, the membrane. The two media contain different
distributions of Na/K/Cl ions. The greater the "stimulation dynamics"
(see below) that first influences
the
outer molecular media - corresponding to sensor 2 in Fig. 2a - and,
subsequently, the inner molecular media - which corresponds to sensor 1
in Fig. 2a - the faster is the Na+ influx and the
shorter
the charging time (which determines the parameter for the relative speed
vm), and the faster is the AP propagation velocity v(ap) in the
neighbouring membrane districts. The signals at the inner and
outer
sides, respectively, of the membrane, correspond to the signal
amplitudes V and W. The velocity
v(ap),
therefore, indirectly generates the invariant time counting pulses ITCP
or the variable vm-proportional time counting pulses VTCP.
These variable VTCP pulses are self-adaptive modulated time pulses that
are correlated to the relative length. As explained in the following
(contrary to the traditional physical sense), no "invariant time" exists
-- only "perceived time" exists. Of essential importance also is the
difference between "stimulation
intensity " whose measurement is determined by the AP frequency and
therefore by the refractory period, and the "stimulation dynamics",
whose measurement is defined by the charge duration of the cell membrane
and therefore also by the speed of the Na+ influx. "Stimulation
dynamics" is not the same as "increase of the stimulation intensity". It
is a measure of the temporal/spatial variation of the position of
the
receptor relative to the position of the stimulus source, and therefore
of the relative speed vm. The
stimulation intensity corresponds to signal amplitudes, from which
vm-adaptive STQ(d) elapse times
Tδ(1,2,3...)
are derived, while the stimulation dynamics is defined by the acquired STQ(v)
parameters.
Fig. 4b and Fig. 4c show the analysis of STQ elapse times
in a molecular/biological model in an easily comprehensible manner. The
results of the analysis are used to generate redundancy-free
auto-adaptive pattern recognition as well as autonomous regulating and
self-organization processes. The organism in the particular example
shown here is forced to distinguish certain types of foreign bodies that
press on its "skin". It must reply with a fast muscle reflex when it
recognizes a pinprick. But it should ignore the stimulus when it
recognizes a blunt object. A continuous vm-adaptive recording of STQ(d)
elapse times by means of VTCP pulses is necessary to do this. The
frequency of these time counting impulses is modulated in accordance
with the STQ(v) parameters of the stimulus dynamics (vm). These STQ(v)
parameters are required for the recording of the STQ(d) elapse times Tδ(1,2,3...) from the signal
amplitude at the current stimulus intensity. The difference between
"stimulation intensity" and "stimulation dynamics" is easily seen in
this example. A stimulus can even show a different intensity if
no temporal-spatial change takes place between signal source and
receptor. A needle in the skin can cause a different sensory pattern
even when its position is not changing if, for example, it is heated.
This sensory pattern is determined by the signal amplitude, and
consequently by the AP frequency and by the STQ(d) quanta. As long as
the needle persists in an invariant position, the AP propagation
velocity is constant, because the membrane charging time is constant
too. During the prick into the skin, there is a "dynamic stimulation",
and the STQ(d) quantization of the signal amplitude is carried out in a
manner that depends on the pricking speed vm. It should be noted that
two temporally displaced signal amplitudes (at the inner and outer
membrane surface) always exist during this dynamic process. The STQ(v)
parameters are derived from this. The AP propagation velocities and the
acquired STQ(d) time patterns are adapted accordingly ("temporal
auto-adaptation").
Fig. 4b
shows the vm-dependent propagation of an AP from a sensory neuron
(receptor) 20 along an
axon to
a synapsis, where a comparison of acquired time sequences takes place
through molecular" covariance analysis". This receptor functions like a
"pressure sensor". If a needle 21 with a certain dynamics
impinges on the outer side of the cell membrane, then this stimulation
causes triggering of AP's 23 as described in Fig. 4a. The AP's
propagate in the axon 22 with a STQ(v)-dependent speed vap. The
sequence (a'.....v') represents the signal amplitude values that are
produced by the pinprick. The sequence begins with the phase transition
at the first threshold value P1, continues over P2, P3, P4 (at which
point the stimulus maximum is attained), and finally to the phase
transitions through P3 and P2. The intensity zones for stimulus
perception are designated with Z1, Z2, Z3 and Z4. The periods t(P1),
t(P2), t(P3), t(P4)......, and the magnitudes of the AP's serve to
identify the particular threshold in which the stimulation intensity is
currently to be found. Their temporal sequence is therefore a type of
"code". AP's are not time counting pulses. Besides their coding
function, they also serve as (indirect) activating
and
deactivating pulses for the recording of STQ(d) elapse times. The actual
vm-dependent
measurement of the STQ elapse times Td(1), Td(2), Td(3), Tw(4) and
Td(4)... (see Fig. 2c), as well as the comparison of these with
previously recorded elapse times, takes place in the synapse 24.
At the presynaptic terminal of the axons, the AP's 23 arrive with
variable velocities vm(n...), according to the dynamics of the needle
prick as well as the measured STQ(v) parameters. This variable arrival
velocity at the synapses is the key to producing the adaptive time
counting impulses VTCP (see Fig. 3c) with vm-modulated frequency ƒscan.
The synapse is separated from the postsynaptic membrane by the "synaptic
cleft", and the postsynaptic membrane, for its part, is interconnected
with other neurons; for instance, to a "motorneuron" 25. This
neuron generates a so-called "excitatory postsynaptic potential" (ESPS)
27 that is approximately proportional to the convergence
probability g. If this EPSP (or, equivalently, the probability density
g) exceeds a certain threshold value, then, in turn, an action potential
AP 28 is triggered. This AP is communicated via motoaxon 26
to the "neuromuscular junction", at which a muscle reflex is triggered.
The incoming AP sequences 23 generate the release of particular
amounts of molecular transmitter substance from their repositories -
tiny spherical structures
in the
synapse, termed "vesicles". In principle, a synapse is a complex
programmable timedata processor and analyzer that empties the contents
of a vesicle into the presynaptic cleft when the recurrence of any prior
recorded synaptic structure is confirmed within a newly recorded key
sequence. The synaptic structures and vesicle motions are generated by
the dynamics (vap) of the AP ionic flux, as well as by its frequency. AP
influx velocities v(ap) correspond to the STQ(v) elapse times, and AP
frequencies correspond to the STQ(d) elapse times. The transmitter
substance is reabsorbed by the synapse, and reused later, whereby the
cycle continues uninterrupted.
We now
present a detailed description of Fig. 4b (referring also to Figs. 4e
and 4f). The ionic influx of the initial incoming AP 23 (a') activates
the spherical structures (vesicles) containing the ACh transmitter
molecules. These molecules are released in the form of a "packet". The
duration of this ACh packaging depends on the dynamics (represented by
the velocity v(ap)) of the AP ionic influx at the presynaptic terminal,
and therefore on the stimulus dynamics (represented by vm) at the
receptor 20. Each subsequent incoming AP, namely b', c'..., in
turn causes neurotransmitter substances in the vesicle to be released
toward the synaptic cleft. Each of the following are elapse time
counting and covariance analyzing characteristics:: the duration of
accumulation of neurotransmitter substance T(t); the velocities v(t)
with which the neurotransmitter substances move in the direction of the
synaptic cleft; the effects induced by the neurotransmitter substances
at the synaptic lattice at the synaptic cleft; the duration of
pore
opening; and so on. By means of AP's acting on synaptic structures, not
only are the actual time counting frequencies ƒscan generated (to be
used in vm-dependent measurement of STQ(d) elapse times as described in
Fig. 2c), but also time patterns are stored and analysed.
If the pattern of a current temporal sequence is recognised by the
synapse as matching an existing stored pattern, a pore opens at the
synaptic lattice, and all of the neurotransmitter content of a vesicle
is released into the subsynaptic cleft. The released transmitter
molecules (mostly ACh) combine at the other side of the cleft with
specific receptor molecules of the sub-synaptic membrane of the coupled
neuron. Thus, a postsynaptic potential (EPSP) is generated, which then
propagates to other synapses, dendrites, or to a "neuromuscular
junction". If the EPSP exceeds a certain amplitude, then it triggers an
action potential (AP) of the described type, which then triggers, for
example, a muscle reflex. If the potential does not reach this
threshold, then the EPSP propagates in the same manner as an EP (i.e. in
an electrotonic manner); an AP is not produced in this case.
Of special significance is the summing property of the subsynaptic
membrane. This characteristic,
termed "temporal facility", results in the summation of amplitudes of
the generated EPSP's, if they arrive in short sequences within certain
time intervals. Each release of neurotransmitter molecules into the
synaptic cleft designates an increased probability density occurring
during the comparison of instantaneous vm-proportionally acquired STQ
time patterns to prior vm-proportionally recorded STQ-time patterns.
Increased probability density causes a higher frequency of transmitter
substance release and therefore a higher summation rate of the EPSP's,
which in turn produces, at a significantly increased rate, postsynaptic
action potentials (AP). Therefore, a postsynaptic AP is effectively a
confirmation signal that flags the fact that isomorphism between a
previously and currently recorded time data pattern has been recognized.
On the basis of this time pattern comparison, the object that caused the
perception at the receptor cell is thereby identified as "needle"; and
the command to "trigger a muscle reflex" is conveyed to the
corresponding muscle fibres.
Parallel and more exact recognition processes are executed by the
central nervous system CNS (i.e. the
brain).
From the sensitive skin-receptor neuron 20, a further axonal
branching 29 is connected via a synapse 30 to a "CNS
neuron". In contrast to the "motorneuron" which actuates the motoric
activity of
the
organism directly, a CNS neuron serves for the conscious recognition of
a receptoric stimulation sequence. An AP 31, produced at the
postsynaptic cell membrane 30, can spread out along dendrites in the
axon 30a, as well as to several other CNS neurons; or,
alternatively, indirectly via CNS neurons to a motorneuron, then on to a
neuromuscular junction.
The parameters controlling the recording of STQ time quanta in the
synapses 25 and 30 can differ with different synaptic
structures. (Indeed, the synaptic structures themselves are generated by
continuous "learning" processes). This explains how it is possible for a
needle prick to be registered by the brain, while eliciting no muscular
response; or how a fast muscle reflex can be produced while a cause is
hardly perceived by the brain. The first case shows a conscious reflex,
the other case an instinctive reflex. The former occurs when the CNS
synapse 30 cannot find enough isomorphic structures (in
contrast to the synapse 25), transmitter molecules are not
released with sufficient frequency, and subsequently no postsynaptic AP
31 and no conscious recognition of the perceived stimulus can
take place. Numerous functions of the central nervous system can be
explained in such a monistic way; as well as phenomena such as
"consciousness" and "subconscious". Generally, auto-adaptive processes
are
deeply
interlaced in organisms, and are therefore extremely complex. In order
to be capable of distinguishing a needle prick from the pressure of a
blunt eraser, essentially more time patterns are necessary; in addition,
more receptors and synapses must be involved in the recognition process.
Fig. 4c illustrates the process by which moderate pressure from a
blunt object (e.g. a conical eraser on a pin) is recognized, resulting
in no muscle reflex. The blunt object 32 presses down with a
certain relative velocity vm onto a series of receptors in neural skin
cells 33, 34, 35, 36 and 37. Several sequences of AP's
39, 40, 41, 42 and 43 are produced after the individual
adjacent receptors (see also Fig. 4b) are stimulated. These action
potentials propagate along the collateral axons 38 with variable
periods t(P1,2,3..) and velocities vap(1..5), which result on the one
hand from the prevailing stimulation intensity, and on the other hand
from the respective stimulation dynamics. Since each receptor stimulus
generates a different pattern of STQ(v) and STQ(d) quanta, various AP
sequences a'.....m' emerge from each axon. All sequences taken together
represent the pattern of STQ elapse times which characterises the
pressure of the eraser on the skin. These variable AP ionic fluxes reach
the synapses 44, 45, 46, 47 and 48, which are
interconnected via the synaptic cleft with the motoneuron 49. As
soon as the currently acquired STQ time data pattern shows a similarity
to a prior recorded STQ time data pattern, each
individual synapse releases the contents of a vesicle into the
subsynaptic cleft. Simultaneously, this produces an EPSP at the
subsynaptic membrane of the neuron. These EPSP potentials are mostly
below the threshold. The required threshold value for the release of an
AP is reached only when a number of EPSP's are summed. This happens only
when a so-called "temporal facilitation" of such potentials occurs, as
described in the previous paragraph.
In the model shown, the individual EPSP's 50, 51, 52, 53 and
54 effect this summing property of the subsynaptic membrane. These
potentials correspond to receptor-specific probability density
parameters g1, g2, g3, g4 and g5, that represent the degree of
isomorphity of time patterns. Simultaneous neurotransmitter release in
several synapses, for example in 45 and 47, causes
particular EPSP's to be summed to a total potential 56, which
represents the sum of the particular probability densities G = g1+g3.
This property of the neurons (i.e. the summing of spatially separated
subliminal EPSP's when release of neurotransmitter substance appears
simultaneously at a number of parallel synapses on the same subsynaptic
membrane) is termed "spatial facilitation".
In the described model case, the summed EPSP 56 does not,
however, reach the marked threshold (gt), and therefore no AP is
produced. Instead, the EPSP propagates in the sub-synaptic membrane
region 49 of the neuron, or in the following motoaxon 55,
respectively, as a passive electrotonic potential (EP). Such
an EP attenuates (in contrast to a self-generating active AP) a few
millimetres along the axon, and therefore has no activating influence on
the neuromuscular junction, and consequently no activating influence on
the muscle. The stimulation of the skin by pressing with the eraser is
therefore not sufficient to evoke a muscle reflex.
It would be a different occurance if the eraser would break off and the
empty pin meet the skin receptors with full force. In this case,
neurotransmitter substances would be released simultaneously in all five
synapses 50, 51, 52, 53 and 54, because the acquired STQ
time patterns Tδ(1,2,3..),
with very high probability, would be similar to those STQ time patterns
Tδ'(1,2,3...
) already stored in the synaptic structures that pertain to the event
"needle prick". The EPSP's would be summed, because of their temporal
and spatial "facilitation", to a supraliminal EPSP 56, and a
postsynaptic AP would be produced that propagates along the motoaxon
55 in a self-regenerating manner (without temporal and spatial
attenuation) up to the muscle, producing a muscle reflex.
As in Fig. 4b, in the present example a recognition process takes place
in the central nervous system (CNS) that proceeds in parallel. From the
skin receptor cells 33, 34, 35, 36 and 37, collateral
axonal branches extend to CNS synapses that are connected to other
neurons 58. Such branches are termed "divergences". The
subdivision of axons into collateral branches in different neural CNS
districts, and the temporal and spatial combination of many postsynaptic
EPSP's, allows conscious recognition of complex perceptions in the brain
(for example, the fact of an eraser pressing onto the skin). Since this
recognition has to take place independent of the production of a muscle
reflex, the sum of individual EPSP's must be supraliminal in the CNS.
Otherwise, no postsynaptic AP - i.e. no signal of confirmation - can be
produced. As an essential prerequisite for this, it is necessary that
auto-adaptive processes have already occurred
which
have formed certain pre-synaptic and sub-synaptic STQ time structures in
the parallel synapses
58. These structures hold information (time sequences; i.e.
patterns) pertaining to similar sensory
experiences (e.g. "objects impinging on the skin" - amongst these, a
conical eraser). Obviously the threshold for causing an AP in the
postsynaptic membrane structure of the ZNS Neurons 58 (and
therefore also in the brain) has to be lower than in the motoneuron
membrane 49 described previously. Therefore also the sum of these
EPSP's must be larger than the sum of the EPSP's g1, g2, g3, g4 and g5.
Isomorphisms of STQ time patterns in the CNS synapses of the brain have
to be more precisely marked out than those in the synapses of
motoneurons, which are only responsible for muscle reflexes.
The
structure of the CNS synapses must be able to discern finer information,
so it must be more subtle. The production of a sub-synaptic AP
represents a confirmation of the fact that a currently acquired Tδ(1,2,3...)
time pattern is virtually isomorphic to a prior recorded reference time
pattern Tδ'(1,2,3...),
which, for example, arose from a former sensory experience with an
eraser impinging at a certain location on the skin. If such a former
experience has not taken place, the consciousness has no physical basis
for the recognition, since the basis for time pattern comparison is
missing. In such a case, therefore, a learning process would first have
to occur. Most of the time, however, sensory experiences of a visual,
acoustic or other type, arising from a variety of receptor stimulation
events, are co-ordinated with the pressure sensing experience.
This explains why CNS structures are extremely intensively interlaced.
CNS neurons, as well as moto-neurons, have up to 5000 coupled synapses,
which are interconnected in a multifarious manner with
receptor neurons and axonal branches. There are complex time data
patterns for lower and higher task sites, which are structured in a
hierarchical manner. We have already described simple Tδ(1,2,3....)
and Tδ'(1,2,3...)
analysis operations. Blood circulation, respiration, co-ordination of
muscle systems, growth, seeing, hearing, speaking, smelling, and so on,
necessitate an extremely large number of synaptic recorded "landscapes"
of the organism's STQ time patterns, produced by a variety of receptors;
and which continually have to be analysed for isomorphism with time
patterns currently being recorded. Accordingly, temporal and motoric
auto-adaptation occurs in deeper and higher hierarchies and at various
levels.
Fig. 4d
illustrate the counterpart to the EPSP (Excitatory Postsynaptic
Potential): the "Inhibitory
Postsynaptic Potential " , or IPSP. As seen in the figure, the IPSP
potentials 61, 62, 63, 64 and 65 at the subsynaptic
membrane 60 are negative compared to the corresponding EPSP's.
IPSP's are produced by a considerable proportion of the synapses to
effect pre-synaptic inhibition instead of activation. The example here
shows an IPSP packet 67 propagating from the motoaxon 66
to a neuromuscular junction (or muscle fibre, respectively) which
prevents this muscle from being activated - even if a supraliminal EPSP
were to reach the same muscle fibre at the same time via a parallel
motoaxon.
Positive EPSP's ion fluxes and negative IPSP's ion fluxes counterbalance
each other. The main function of the IPSP's is to enable co-ordinated
and homogeneous changes of state in the organism, e.g. to enable exact
timing of motion sequences. In order to ensure, for example, a constant
arm swing, it is necessary to activate the bicep muscles, which then
flex the elbow with the aid of EPSP's; but to inhibit the antagonistic
tricep muscles (which extend the elbow) with the aid of IPSP's.
Antagonist muscles must be inhibited via so-called "antagonistic
motoneurons", while the other muscle is activated via "homonym
motoneurons". The complex synergism of excitatory (EPSP) synapses and
inhibitory (IPSP) synapses act like a feedback system (servoloop) and
enables optimal timing and efficiency in the organism. One can compare
this process with a servo-drive, or with power-steering, which ensures
correct co-ordination and execution of current motion through
data-supported operations and controls. If data are missing, the
servoloop collapses. Disturbances in a molecular biological servoloop
that is supported by STQ time data structures lead to tetanic twitches,
arbitrary contractions, chaotic cramps and so on.
From the point of view of cybernetics, each excitatory synapse generates
a "motoric impulse" (EPSP), while each inhibitory synapse generates a
"brake impulse" (IPSP). The continued tuning of the complicated
servoloops, and the balance which results from continuous comparison of
prior sensory experiences (the stored reference time patterns) with
current sensory experiences (the time patterns currently being
recorded), creates "perfect timing" in the organism.
Fig. 4e shows the basic construction of a synapse. Axon 68
ends at the pre-synaptic terminal 69, which is also termed
"bouton". The serial incoming AP's cause the vesicles to be filled with
neurotransmitter molecules. When the filling process is finished, the
vesicles begin to move in the direction of the pre-synaptic lattice
71. If a currently acquired time pattern is approximately isomorphic
to an existing time pattern (see also Fig. 4b), then a small canal opens
at an attachment site on the lattice, which releases the
entire contents of the vesicle into the narrow synaptic cleft 72.
This process is termed "exocytosis". The
sub-synaptic neural membrane 73 supports specific molecular
receptors 73a, to which the released transmitter molecules bind
themselves. For a certain period, a pore opens, through which the
transmitter substance diffuses. The conductivity of the postsynaptic
membrane increases and the EPSP (following postsynaptic depolarisation)
is triggered. The duration of opening of the pores and the recognition
of complementary receptors by the molecules are likewise determined by
auto-adaptive processes and evaluation of STQ time pattern structures.
However, these molecular processes represent deeper sub-phenomena in
comparison to synaptic processes. Structures for temporal and motoric
auto-adaptation, which depend on quantization of STQ-elapse times, also
exist at the molecular and atomic levels.
Fig. 4f shows the filling of a vesicle 70 with
neurotransmitting substances, and its subsequent motion towards a
pre-synaptic dense projection at the lattice 71. The start of the
filling process 74 can be seen as the activation of a stopwatch.
The rate v(t) of the filling is proportional to the dynamics of the AP
ionic flux into the synapse. The periods T(t...) of the filling follow
the periods t(P1,P2,...) of the arriving AP's; these times, therefore,
represent vm-adaptive quantized STQ(d) elapse times Tδ(1,2,3...).
The direction of filling is shown at 75. The direction of motion
of a vesicle is shown at 76. If the current velocity v(t), the
duration of the vesicle packaging T(t), the quantity of transmitter
molecules, the current vesicle motion and other currently significant
STQ parameters have characteristics which correlate to an existing
synaptic STQ structure, then a filled vesicle binds itself onto an
"attachment site" 77 at the lattice. Ca++ ions flow into the
synapse, a pore at the para-crystalline vesicle lattice opens, and the
entire molecular neurotransmitter content is released into the synaptic
cleft 72. At the postsynaptic membrane
of the target neuron, these molecules are fused with specific receptor
molecules. Such receptors have verification tasks. They prevent foreign
transmitter substances (that originate from other synapses) from
producing wrong ESPS's at this neuron.
To complete the discussion of Fig. 4, we relate the descriptions of
Figs. 4a, 4b, 4e and 4f to the STQ-configurations of Figs. 3a - g. For
argument's sake, we assume once again that a pinprick impinges onto a
receptor cell (see also Fig. 4b).
The IP sequences shown in Fig. 3a correspond to the AP's 23 which
are produced by stimulating a receptor cell 20 with a needle
21. Their periods t(P1), t(P2),... serve to classify the respective
zones of stimulation intensity (P1, P2...) or perception intensity (Z1,
Z2... ). Each AP 23, arriving into a synapse 69, activates the
adaptive quantization of STQ(d) elapse times, depending on the velocity
vap of the propagation of the AP along the axon. Elapse timing with
modulated time base is triggered as soon as a vesicle begins to fill.
Finished filling (packaging) signifies "elapse timing stop, STQ(d)-
quantum recorded". The elapse times Td(1),
Td(2),
Td(3),
Td(4)....
thus recorded generate the significant synaptic structures. Invariant
time counting pulses ITCP (see Fig. 3b) with frequency fscan correspond
to constant axonal AP propagation with velocity vap, if no dynamic
stimulus appears at the skin receptor cell (for example, if a needle
remains in a fixed position and generates a constant stimulation
intensity). In this case, the receptor membrane senses no relative speed
vm; the AP's propagate with constant velocity vap along the axon 22; and
the synapse quantizes the STQ(d) elapse times with invariant time
counting frequency fscan.
Time counting pulses VTCP (see Fig. 3c) with variable frequency ƒscan
are then applied, if dynamic stimulation affects the receptor. The AP's
propagate along the axon with STQ(v)-dependent velocities vap(n...),
modulated by the variable dynamics vm(n...) which are measured as an
STQ(v) parameter by the membrane. Adaptive alteration of all of the
following processes occurs in a similar manner: the variation of time
counting periods t(P1... .n) corresponding to the points 2.1, 3.1, 4.1
in Fig. 3c; the velocities v(t....) of AP ionic flux into the synapse;
the vesicle filling times T(t...); the amounts of transmitter molecules
contained in the vesicles; the motion of these molecules in the
direction of the vesicle lattice; the structure of this lattice; and
many other parameters of the presynaptic and subsynaptic structures.
A synapse has features that enable the conversion of the AP influx
dynamics into vap-proportional molecular changes of states. This is like
the variable VTCP time counting pulses seen in Fig. 3c. The process can
be compared with variable water pressure driving a turbine, through
which a generator produces variable frequencies depending on pressure
and water speed: higher water pressure is akin to higher stimulation
dynamics vm at the receptor, higher AP propagation velocity vap along
the axon, and higher VTCP time pulse frequency ƒscan in the synapse
(which in turn affects not only the rate v(t) with which vesicles are
filled, but also many other synaptic parameters). According to these
processes, the STQ(d) time sequence Td(1,
2, 3, 4...) is recorded in the synapse with vm-modulated time counting
frequencies ƒscan(1,2,3...); as a consequence, the physical structure of
the synapse is determined by this time sequence.
Fig. 3d shows a currently acquired time data sequence 32 30 22 23 20
that is equivalent to the recorded time pattern Tδ(1,2,3..),
and which leaves a specific molecular biological track in the synapse
24. The prior acquired time data sequence 30 29 22 24 19 in Fig. 3e
corresponds to the synaptic structure that has been "engraved" through
frequent repetition of particular stimulation events and time patterns Tδ'(1,2,3...).The
manifested synaptic Td' structure can be considered also as a bootstrap
sequence that was generated
by
continuous learning processes and perception experiences, and which, for
example, serves as a
reference pattern for the event "pinprick". If a newly acquired Td
bootstrap sequence – which is given by
the
current properties of the vesicle filling, as well as other significant
time dependent parameters - approximately keeps step with this existing
Tδ'
(bootstrap sequence (or with a part of it), then
"covariance" is acknowledged in the synaptic structure. This opens a
vesicle attachment site at the
synaptic lattice and results in the release of all transmitter molecules
that are contained in a vesicle,
whereupon an EPSP is generated at the sub-synaptic membrane 25.
The potential of an EPSP
corresponds to the probability density parameters shown in Fig. 3f,
which are significant for the
currently evaluated covariance. If such "probability density parameters"
sum within a certain time
interval to a certain threshold potential 27, an AP 26 is
produced. This AP serves as confirmation of the event "pin recognized",
and produces a muscle reflex.
The comparison of the current elapse time pattern with prior recorded
elapse time patterns, as shown in Fig. 3c, takes place continuously in
the synapses. Each recognized covariance of a new time sequence, that is
recorded by "temporal auto-adaptation", sets a type of "servoloop
mechanism" in motion. It initiates a process that we term "motoric
auto-adaptation", and which can be understood as the actual "motor" in
biological chemical organisms, or life forms, respectively. Structures
of temporal and motoric auto-adaptation, which are based on STQ
quantization, exist also at the lowest molecular level. Without
elapse time-supported servoloops, co-ordinated change in biological
systems would be
impossible. This applies especially to the motion of proteins; to the
recognition and replication of the genetic code; and to other basic life
processes. The creation of higher biological/chemical order and complex
systems such as synapses or neurons presupposes the existence of an STQ
quantization molecular sub-structure, from which simple acknowledgement
and self-organization processes at a lower level derive. Indeed, there
are innumerable hierarchies of auto-adaptive phenomena on various
levels. Simple phenomena on a molecular level also include: fusion of
receptor molecules; the formation of pores, ion canals and sub-axonal
transportation structures (microtubules); and the formation of new
synapses and axonal branchings.
By this token, recognition of stimulation signal sequences by synaptic
time pattern comparison (as an involuntary reflex or as a conscious
perception), as discussed in the description of Figs.
4a - c, is an STQ-epiphenomenon. Each such auto-adaptive
STQ-epiphenomenon, for its part, is superimposed from STQ-epiphenomena
of higher rankings; for example, the analysis of complex "time
landscapes" in order to find isomorphism. STQ-epiphenoma such as
regulation of blood circulation, body temperature, respiration, the
metabolism, seeing, hearing, speaking, smell, the co-ordination of
motion, and so on, are for their parts superimposed from STQ-scenarios
of higher complexity, including consciousness, thought, free will,
conscious action, as well as an organism's sensation of time. In all
these cases, the central nervous system looks after convergent time
patterns that are placed like pieces of a jigsaw puzzle into an
integrated total sensory scenario.
If, in any hierarchy, within a certain "latency time" (i.e. time limit)
and despite intensive "searching", no time subpattern covariant with the
STQ time pattern can be found, then the organism displays chaotic
behaviour. This behaviour restricts itself to that synaptic part in
which the non-convergence has appeared. As soon as a covariant time
pattern is found, the co-ordinated process of temporal and motoric
auto-adaptation (and auto-emulation) resumes. (This can be likened to
servo-steering that has collapsed for a short time.) However, the
"chaotic behaviour" is itself quantized as an STQ time pattern, and is
recorded by the affected synapses in such a manner that no
neurotransmitter substance release occurs despite arriving AP's. Via
subaxonal transportation structures (i.e. the microtubules) such
information streams back borne on transmitter molecules which travel in
the inverse direction along the axon.
Microtubules are used to generate new synapses and synaptic connections
at the neurons and neural
networks in which a collapse of an auto-adaptation process has occurred.
The production of new
synapses proceeds to the generation of dendrites; i.e., axonal branches
that carry processing information from
neurons. In this way the auto-adaptive neural feedback mechanism
regenerates itself, and the time
pattern that was acquired during the short termed "chaotic behaviour"
becomes a new reference basis for the recognition of future events.
Thus, the CNS learns to record new events and experiences;
and
learns to evaluate time patterns which were unknown previously.
send your e-mail to: info@sensortime.com
you are the visitor Nr info@sensortime.com